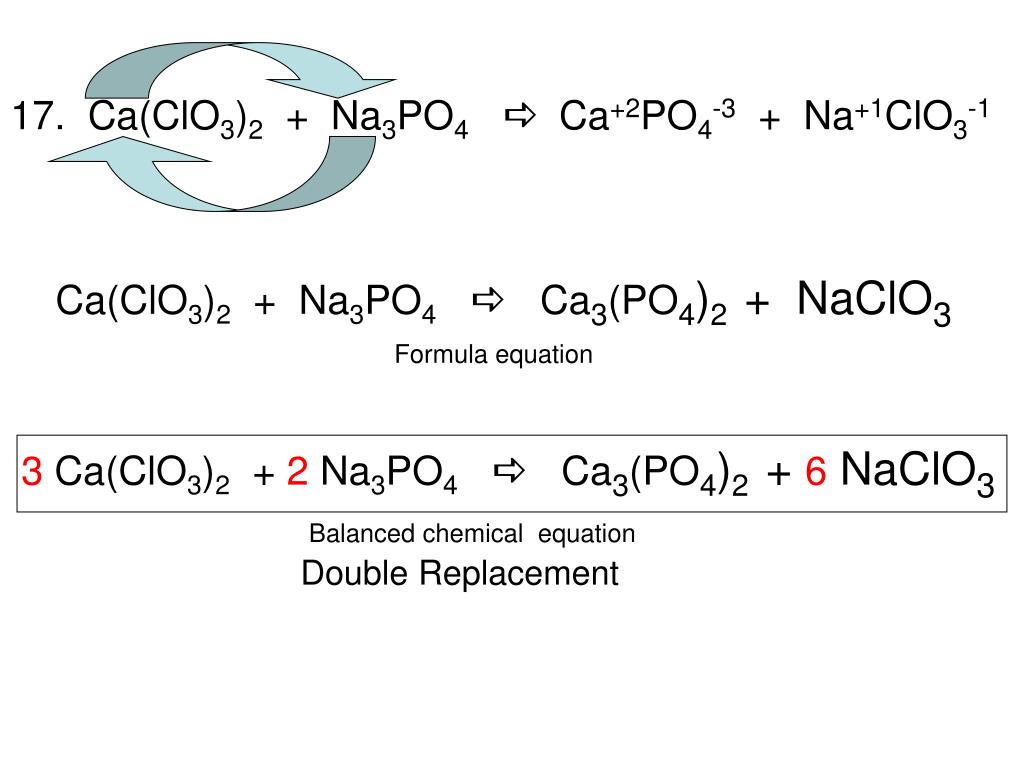
OneClass: I have started this problem but now I am stuck. Please show all work on how the answer is ...

SOLVED: Please express the following chemical reactions as their chemical equilibrium formula. Give the balanced equations and give the chemical equilibrium formula 1.) AgNO3(aq) K2CrO4(aq) = Ag2NO3 KNO3 2.) CuCl2(aq) Pb(NO3)2(aq) Cu(NO3)2
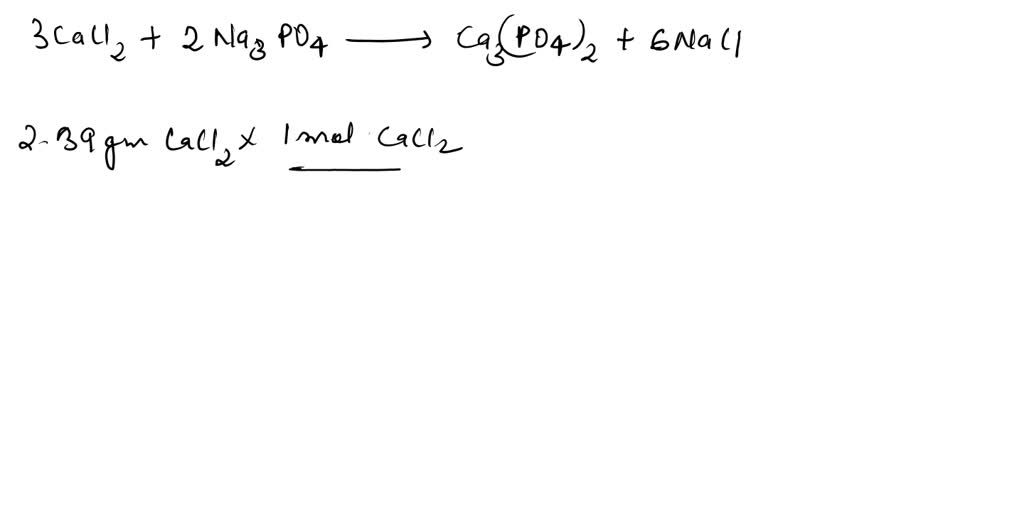
SOLVED: 3 CaCl2(aq) + 2 Na3PO4(aq) -> Ca3(PO4)2(s) + 6 NaCl(aq) 1,50 M 250,0 mL CaCl2 solution is reacted with excess Na3PO4 solution. Mass of the solid product is 36,7 g. What

CaCl2+Na3PO4=Ca3(PO4)2 + NaCl Balanced Equation||Calcium chloride+Sodium phosphate Balanced Equation - YouTube



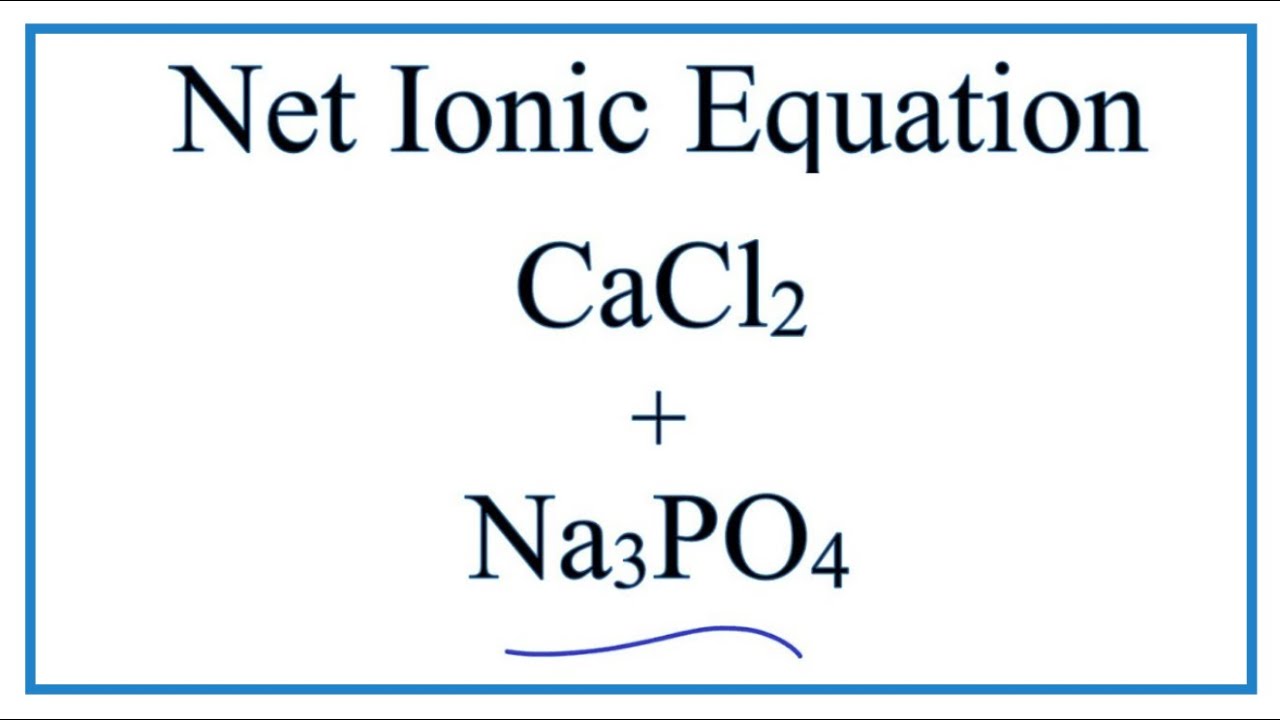
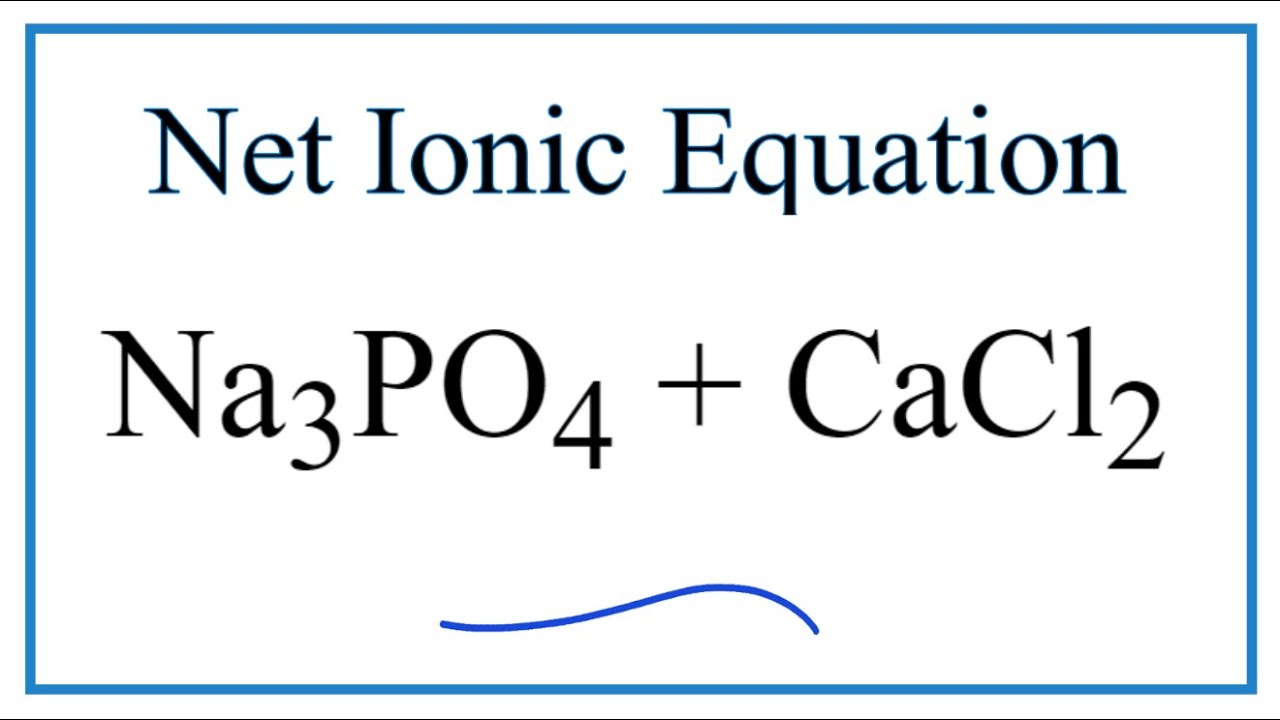

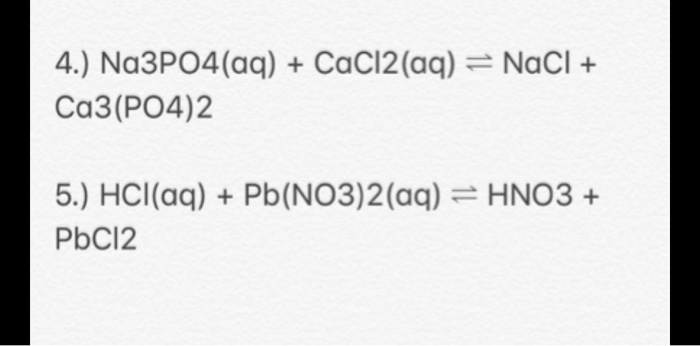



![ANSWERED] Calcium chloride reacts with sodium phosp... - Physical Chemistry ANSWERED] Calcium chloride reacts with sodium phosp... - Physical Chemistry](https://media.kunduz.com/media/sug-question/raw/57553417-1659623475.8752673.jpeg)